Alaproclate
 | |
| Clinical data | |
|---|---|
| Routes of administration |
Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C13H18ClNO2 |
| Molar mass | 255.74 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
|
| |
Alaproclate (developmental code name GEA-654) is a drug that was being developed as an antidepressant by the Swedish pharmaceutical company Astra AB (now AstraZeneca) in the 1970s. It acts as a selective serotonin reuptake inhibitor (SSRI), and along with zimelidine and indalpine, was one of the first of its kind. Development was discontinued due to the observation of liver complications in rodent studies. In addition to its SSRI properties, alaproclate has been found to act as a non-competitive NMDA receptor antagonist, but does not have discriminative stimulus properties similar to phencyclidine.
Synthesis
Method for Treatment of Senile Dementia:
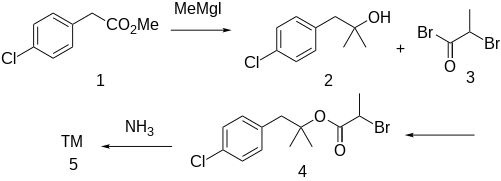
The Grignard reaction between methyl 4-chlorophenylacetate [52449-43-1] (1) with methylmagnesium iodide gives 1-(4-chlorophenyl)-2-methyl-2-propanol [5468-97-3] (2). Acylation with 2-bromopropionyl bromide [563-76-8] (3) gives [1-(4-Chlorophenyl)-2-methylpropan-2-yl] 2-bromopropanoate, CID:13695101 (3). Displacement of halogen with ammonia leads to alaproclate (4).
See also
| AMPAR |
|
|---|---|
| KAR |
|
| NMDAR |
|