Amenamevir
 |
|
| Trade names |
Amenalief |
| Other names |
ASP-2151, ASP2151 |
| ATC code |
|
|
| Legal status |
- In general: ℞ (Prescription only)
|
|
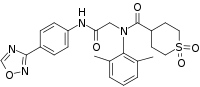
N-(2,6-Dimethylphenyl)-N-[2-[4-(1,2,4-oxadiazol-3-yl)anilino]-2-oxoethyl]-1,1-dioxothiane-4-carboxamide
|
| CAS Number |
|
|
PubChem CID
|
|
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
|
| Formula |
C24H26N4O5S
|
| Molar mass |
482.56 g·mol−1
|
| 3D model (JSmol) |
|
Cc1cccc(C)c1N(CC(=O)Nc1ccc(-c2ncon2)cc1)C(=O)C1CCS(=O)(=O)CC1
|
InChI=1S/C24H26N4O5S/c1-16-4-3-5-17(2)22(16)28(24(30)19-10-12-34(31,32)13-11-19)14-21(29)26-20-8-6-18(7-9-20)23-25-15-33-27-23/h3-9,15,19H,10-14H2,1-2H3,(H,26,29) Key:MNHNIVNAFBSLLX-UHFFFAOYSA-N
|
Amenamevir (trade name Amenalief) is an antiviral drug used for the treatment of shingles (herpes zoster).
It acts as an inhibitor of the zoster virus's helicase–primase complex. Amenamevir was approved in Japan for the treatment of shingles in 2017.

