
Casopitant
Подписчиков: 0, рейтинг: 0
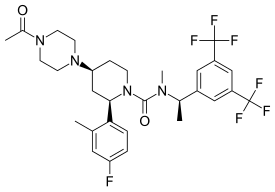 | |
| Clinical data | |
|---|---|
| Other names | GW679769; GW-679769; (2R,4S)-4-(4-Acetylpiperazin-1-yl)-N-{(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethyl}-2-(4-fluoro-2-methylphenyl)-N-methylpiperidine-1-carboxamide |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C30H35F7N4O2 |
| Molar mass | 616.625 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Casopitant (INN), former tentative trade names Rezonic (U.S.) and Zunrisa (Europe), is an NK1 receptor antagonist which was undergoing research for the treatment of chemotherapy-induced nausea and vomiting. It was under development by GlaxoSmithKline. In July 2008, the company filed a marketing authorisation application with the European Medicines Agency. The application was withdrawn and development was discontinued in September 2009 because GlaxoSmithKline decided that further safety assessment was necessary. However, a 2022 review listed casopitant as under development as a potential novel antidepressant for the treatment of major depressive disorder, with a phase 2 clinical trial having been completed.