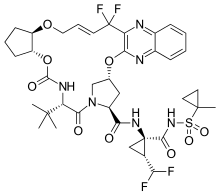
Glecaprevir
 |
|
| Trade names |
Mavyret (combination with pibrentasvir) |
| Other names |
ABT-493 |
Routes of
administration |
By mouth |
| ATC code |
|
|
| Protein binding |
97.5% |
| Metabolism |
CYP3A |
| Elimination half-life |
6 hours |
| Excretion |
Faeces (92.1%), urine (0.7%) |
|
(3aR,7S,10S,12R,21E,24aR)-7-tert-Butyl-N-{(1R,2R)-2-(difluoromethyl)-1-[(1-methylcyclopropane-1-sulfonyl)carbamoyl]cyclopropyl}-20,20-difluoro-5,8-dioxo-2,3,3a,5,6,7,8,11,12,20,23,24a-dodecahydro-1H,10H-9,12-methanocyclopenta[18,19] [1,10,17,3,6]trioxadiazacyclonon adecino[11,12-b]quinoxaline-10-carboxamide
|
| CAS Number |
|
|
PubChem CID
|
|
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
|
| Formula |
C38H46F4N6O9S
|
| Molar mass |
838.87 g·mol−1
|
| 3D model (JSmol) |
|
CC(C)(C)[C@@H]1NC(=O)O[C@@H]2CCC[C@H]2OC/C=C/C(F)(F)c2nc3ccccc3nc2O[C@@H]2C[C@@H](C(=O)N[C@]3(C(=O)NS(=O)(=O)C4(C)CC4)C[C@H]3C(F)F)N(C2)C1=O
|
InChI=1S/C38H46F4N6O9S/c1-35(2,3)28-32(50)48-19-20(17-24(48)30(49)46-37(18-21(37)29(39)40)33(51)47-58(53,54)36(4)14-15-36)56-31-27(43-22-9-5-6-10-23(22)44-31)38(41,42)13-8-16-55-25-11-7-12-26(25)57-34(52)45-28/h5-6,8-10,13,20-21,24-26,28-29H,7,11-12,14-19H2,1-4H3,(H,45,52)(H,46,49)(H,47,51)/b13-8+/t20-,21+,24+,25-,26-,28-,37-/m1/s1 Key:MLSQGNCUYAMAHD-ITNVBOSISA-N
|
Glecaprevir (INN,) is a hepatitis C virus (HCV) nonstructural (NS) protein 3/4A protease inhibitor that was identified jointly by AbbVie and Enanta Pharmaceuticals. It is being developed as a treatment of chronic hepatitis C infection in co-formulation with an HCV NS5A inhibitor pibrentasvir. Together they demonstrated potent antiviral activity against major HCV genotypes and high barriers to resistance in vitro.
On 19 December 2016, AbbVie submitted a new drug application to the U.S. Food and Drug Administration for the glecaprevir/pibrentasvir (trade name Mavyret) regimen for the treatment of all major genotypes (1–6) of chronic hepatitis C. On 3 August 2017 the FDA approved the combination for hepatitis C treatment. In Europe, it was approved on 17 August 2017 for the same indication, under the trade name Maviret.
See also

