
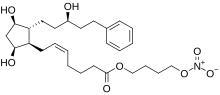
Latanoprostene bunod
Подписчиков: 0, рейтинг: 0
 | |
| Clinical data | |
|---|---|
| Trade names | Vyzulta |
| Other names | BOL-303259-X |
| AHFS/Drugs.com | Multum Consumer Information |
| License data | |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ECHA InfoCard | 100.251.571 |
| Chemical and physical data | |
| Formula | C27H41NO8 |
| Molar mass | 507.624 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Latanoprostene bunod (trade name Vyzulta) is an ophthalmic drug approved in the United States in 2017 for the reduction of intraocular pressure in patients with open-angle glaucoma or ocular hypertension. It targets the trabecular meshwork directly.