
Marimastat
Подписчиков: 0, рейтинг: 0
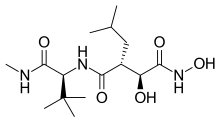 | |
| Clinical data | |
|---|---|
| Routes of administration |
By mouth |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C15H29N3O5 |
| Molar mass | 331.413 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
|
| |
Marimastat was a proposed antineoplastic drug developed by British Biotech. It acted as a broad-spectrum matrix metalloproteinase inhibitor.
Marimastat performed poorly in clinical trials, and development was terminated. This may be, however, a result of targeting cancer at too late of a stage. This is supported by the fact that MMP inhibitors have more recently been shown in animal models to be more effective in earlier stages of cancers. (Effects of angiogenesis inhibitors on multistage carcinogenesis in mice. Science 284, 808-812. Bergers, G., Javaherian, K., Lo, K.-M., Folkman, J., and Hanahan, D. (1999)).