
Mazapertine
| |
| Names | |
|---|---|
|
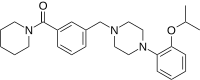
Preferred IUPAC name
(Piperidin-1-yl){3-[(4-{2-[(propan-2-yl)oxy]phenyl}piperazin-1-yl)methyl]phenyl}methanone | |
| Other names
RWJ-37796
| |
| Identifiers | |
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
|
PubChem CID
|
|
| UNII | |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C26H35N3O2 | |
| Molar mass | 421.585 g·mol−1 |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Mazapertine (RWJ-37796) is an antipsychotic agent that was developed by Johnson & Johnson but never marketed. It exerts its pharmacological effect through affinity for dopamine D2, serotonin 5-HT1A, and α1-adrenergic receptors.
Mazapertine is safe and well tolerated when administered orally.
Analogs of mazapertine with conformational restriction have been prepared and have greater affinity for the 5-HT1A receptor.
Synthesis
Alkylation of 2-Nitrophenol [88-75-5] (1) with isopropyl bromide gives 2-isopropoxynitrobenzene [38753-50-3] (2). Catalytic hydrogenation of nitro group gives 2-isopropoxyaniline [29026-74-2] (3). Intermolecular ring formation of this aniline with nornitrogen mustard [334-22-5] yields 1-(2-isopropoxyphenyl)piperazine [54013-91-1] (4).
Amide formation of 3-(chloromethyl)benzoyl chloride [63024-77-1] (5) with piperidine gives 1-[3-(chloromethyl)benzoyl]piperidine [148583-64-6] (6).
Ex 1: The last step is the convergent synthesis between the above two arms of the synthesis to afford the alkylation product mazapertine (7).
