
Rumenic acid
| |
| Names | |
|---|---|
|
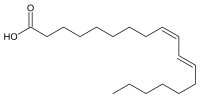
Preferred IUPAC name
(9Z,11E)-Octadeca-9,11-dienoic acid | |
| Other names
Bovinic acid; C9-T11 acid
| |
| Identifiers | |
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider |
|
| KEGG |
|
|
PubChem CID
|
|
| UNII | |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C18H32O2 | |
| Molar mass | 280.452 g·mol−1 |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Rumenic acid, also known as bovinic acid, is a conjugated linoleic acid (CLA) found in the fat of ruminants and in dairy products. It is an omega-7 trans fatty acid. Its lipid shorthand name is cis-9, trans-11 18:2 acid. The name was proposed by Kramer et al. in 1998. It can be considered as the principal dietary form, accounting for as much as 85-90% of the total CLA content in dairy products.
Biosynthesis and biotransformations
Rumenic acid is produced from vaccenic acid by the action of unsaturase enzymes. Rumenic acid is converted back to vaccenic acid en route to stearic acid
Further reading
F. Destaillats; E. Buyukpamukcu; P.-A. Golay; F. Dionisi & F. Giuffrida (2005). "Letter to the Editor: Vaccenic and Rumenic Acids, A Distinct Feature of Ruminant Fats". Journal of Dairy Science. 88 (449): 449. doi:10.3168/jds.S0022-0302(05)72705-3. PMID 15653508.