
Abrocitinib
 | |
| Clinical data | |
|---|---|
| Trade names | Cibinqo |
| Other names | PF-04965842 |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Routes of administration |
By mouth |
| Drug class | Janus kinase inhibitor |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 2.8–5.2 h |
| Excretion | 1.0–4.4% unchanged in urine |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEMBL | |
| ECHA InfoCard | 100.251.498 |
| Chemical and physical data | |
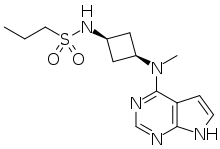
| Formula | C14H21N5O2S |
| Molar mass | 323.42 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Abrocitinib, sold under the brand name Cibinqo, is a medication used for the treatment of atopic dermatitis (eczema). It is a Janus kinase inhibitor and it was developed by Pfizer. It is taken by mouth.
The most common side effects include nausea (feeling sick), headache, acne, herpes simplex (viral infection of the mouth or the genitals), increased levels of creatine phosphokinase in the blood (an enzyme released into the blood when muscle is damaged), vomiting, dizziness and pain in the upper belly.
Abrocitinib was approved for medical use in the European Union in December 2021, and in the United States in January 2022.
Medical uses
In the EU, abrocitinib is indicated for the treatment of moderate-to-severe atopic dermatitis in adults who are candidates for systemic therapy.
In the US, abrocitinib is indicated for the treatment of people twelve years of age and older with refractory, moderate-to-severe atopic dermatitis whose disease is not adequately controlled with other systemic drug products, including biologics, or when use of those therapies is inadvisable.
Side effects
The most common adverse effects in studies were upper respiratory tract infection, headache, nausea, and diarrhea.
Pharmacology
Mechanism of action
It is a selective inhibitor of the enzyme janus kinase 1 (JAK1).
Pharmacokinetics
Abrocitinib is quickly absorbed from the gut and generally reaches highest blood plasma concentrations within one hour. Only 1.0 to 4.4% of the dose are found unmetabolized in the urine.
History
- April 2016: initiation of Phase 2b trial
- December 2017: initiation of JADE Mono-1 Phase 3 trial
- May 2018: Results of Phase 2b trial posted
- October 2019: Results of Phase 3 trial presented
- June 2020: Results of second Phase 3 trial published
Society and culture
Legal status
In October 2021, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Cibinqo, intended for the treatment of atopic dermatitis. The applicant for this medicinal product is Pfizer Europe MA EEIG. In December 2021, the European Commission approved abrocitinib for the treatment of atopic dermatitis.
In January 2022, the US Food and Drug Administration (FDA) approved abrocitinib for adults with moderate-to-severe atopic dermatitis.
External links
- "Abrocitinib". Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT03349060 for "Study to Evaluate Efficacy and Safety of PF-04965842 in Subjects Aged 12 Years And Older With Moderate to Severe Atopic Dermatitis (JADE Mono-1)" at ClinicalTrials.gov
- Clinical trial number NCT03575871 for "Study Evaluating Efficacy and Safety of PF-04965842 in Subjects Aged 12 Years And Older With Moderate to Severe Atopic Dermatitis (JADE Mono-2)" at ClinicalTrials.gov
- Clinical trial number NCT03720470 for "Study Evaluating Efficacy and Safety of PF-04965842 and Dupilumab in Adult Subjects With Moderate to Severe Atopic Dermatitis on Background Topical Therapy (JADE Compare)" at ClinicalTrials.gov
| Chemokine |
|
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CSF |
|
||||||||||||
| Interferon |
|
||||||||||||
| Interleukin |
|
||||||||||||
| TGFβ |
|
||||||||||||
| TNF |
|
||||||||||||
| Others |
|
||||||||||||