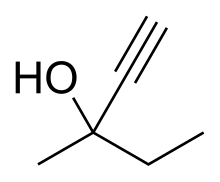
Methylpentynol (Methylparafynol, Dormison, Atemorin, Oblivon) is a tertiary hexynol with hypnotic/sedative and anticonvulsant effects and an exceptionally low therapeutic index. It was discovered by Bayer in 1913 and was used shortly thereafter for the treatment of insomnia, but its use was quickly phased out in response to newer drugs with far more favorable safety profiles.
The drug was marketed again in the United States, Europe and elsewhere from 1956 well into the 1960s as a rapid-acting sedative. The drug was quickly overshadowed at that point by benzodiazepines and is no longer sold anywhere.
Synthesis
Methylpentynol is prepared by reaction of butanone (MEK) with sodium acetylide. This reaction must be done in anhydrous conditions and in an inert atmosphere.
Applications
As building block in the synthesis of:
-
Phthalofyne (1,2-Benzenedicarboxylic acid, mono(1-ethyl-1-methyl-2-propynyl) ester) [131-67-9]
-
Anansiol (1-ethyl-1-methylprop-2-ynyl carbamate) [302-66-9]
- Bason ( 2-Bromoethynyl-2-butanol) [2028-52-6]
See also
|
|
|---|
| By consumption |
|
Primary
alcohols (1°) |
| Methanol |
|
| Ethanol |
|
| Butanol |
|
Straight-chain
saturated
C1 — C9
|
|
Straight-chain
saturated
C10 — C19
|
|
Straight-chain
saturated
C20 — C29
|
|
Straight-chain
saturated
C30 — C39
|
|
Straight-chain
saturated
C40 — C49
|
|
|
Secondary
alcohols (2°) |
|
Tertiary
alcohols (3°) |
|
| Hydric alcohols |
| Monohydric alcohols |
|
| Dihydric alcohols |
|
| Trihydric alcohols |
|
| Polyhydric alcohols |
|
|
| Amyl alcohols |
|
| Aromatic alcohols |
|
Saturated
fatty alcohols
|
|
Branched and
unsaturated
fatty alcohols
|
|
| Sugar alcohols |
| C1 — C7
|
|
Deoxy sugar
alcohols |
|
Cyclic sugar
alcohols |
|
| Glycylglycitols |
|
|
| Terpene alcohols |
Monoterpene
alcohols |
|
Sesquiterpene
alcohols |
|
Diterpene
alcohols |
|
|
| Dialcohols |
|
| Trialcohols |
|
| Sterols |
|
| Fluoroalcohols |
|
| Preparations |
|
| Reactions |
|
|
|
|
|---|
| Alcohols |
|
| Barbiturates |
|
| Benzodiazepines |
|
| Carbamates |
|
| Flavonoids |
|
| Imidazoles |
|
|
Kava constituents |
|
| Monoureides |
|
| Neuroactive steroids |
|
| Nonbenzodiazepines |
|
| Phenols |
|
| Piperidinediones |
|
| Pyrazolopyridines |
|
| Quinazolinones |
|
|
Volatiles/gases
|
|
| Others/unsorted |
- 3-Hydroxybutanal
- α-EMTBL
- AA-29504
- Alogabat
-
Avermectins (e.g., ivermectin)
-
Bromide compounds (e.g., lithium bromide, potassium bromide, sodium bromide)
- Carbamazepine
- Chloralose
- Chlormezanone
- Clomethiazole
- Darigabat
- DEABL
- Deuterated etifoxine
-
Dihydroergolines (e.g., dihydroergocryptine, dihydroergosine, dihydroergotamine, ergoloid (dihydroergotoxine))
- DS2
- Efavirenz
- Etazepine
- Etifoxine
-
Fenamates (e.g., flufenamic acid, mefenamic acid, niflumic acid, tolfenamic acid)
- Fluoxetine
- Flupirtine
- Hopantenic acid
- KRM-II-81
- Lanthanum
- Lavender oil
-
Lignans (e.g., 4-O-methylhonokiol, honokiol, magnolol, obovatol)
- Loreclezole
- Menthyl isovalerate (validolum)
- Monastrol
- Niacin
- Niacinamide
- Org 25,435
- Phenytoin
- Propanidid
- Retigabine (ezogabine)
- Safranal
- Seproxetine
- Stiripentol
-
Sulfonylalkanes (e.g., sulfonmethane (sulfonal), tetronal, trional)
-
Terpenoids (e.g., borneol)
- Topiramate
-
Valerian constituents (e.g., isovaleric acid, isovaleramide, valerenic acid, valerenol)
|
|