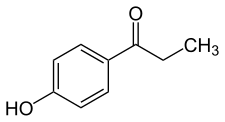
Paroxypropione, also known as paraoxypropiophenone, is a synthetic nonsteroidal estrogen which has been used medically as an antigonadotropin in Spain and Italy but appears to no longer be marketed. It was first synthesized in 1902. The antigonadotropic properties of the drug were discovered in 1951 and it entered clinical use shortly thereafter.
Pharmacology
Pharmacodynamics
Paroxypropione is closely related structurally to p-hydroxybenzoic acid and parabens such as methylparaben, and also bears a close resemblance to diethylstilbestrol (which, in fact, produces paroxypropione as an active metabolite) and alkylphenols like nonylphenol, all of which are also estrogens. The drug possesses relatively low affinity for the estrogen receptor and must be given at high dosages to achieve significant estrogenic and antigonadotropic effects, for instance, 0.8 to 1.6 g/day. It possesses 0.1% of the estrogenic activity and less than 0.5% of the antigonadotropic potency of estrone.
Chemistry
Synthesis
The highest reported yield, approximately 96%, is from the between phenol and propionyl chloride. The mechanism is likely to involve initial esterification to give phenyl propionate, which then undergoes a Fries rearrangement.
Derivatives
Paroxypropione is a precursor in the chemical synthesis of diethylstilbestrol and dienestrol.
Society and culture
Names
Brand names Frenantol, Frenormon, Hypophenon, Paroxon, Possipione, Profenone, numerous others; former developmental code name NSC-2834), also known as paroxypropiophenone (P.O.P.) or 4'-hydroxypropiophenone.
Research
Paroxypropione was studied and used in the treatment of breast cancer.
Further reading
-
Gustavo RP (July 1958). "[Anti-gonadotropic action of possipione]" [Anti-gonadotropic action of possipione]. Quaderni di Clinica Ostetrica e Ginecologica (in Italian). 13 (7): 307–15. PMID 13579130.
|
|
|---|
| Estrogens |
|
ER agonists |
-
Steroidal: Alfatradiol
- Certain androgens/anabolic steroids (e.g., testosterone, testosterone esters, methyltestosterone, metandienone, nandrolone esters) (via estrogenic metabolites)
- Certain progestins (e.g., norethisterone, noretynodrel, etynodiol diacetate, tibolone)
- Clomestrone
- Cloxestradiol acetate
- Conjugated estriol
- Conjugated estrogens
- Epiestriol
- Epimestrol
- Esterified estrogens
-
Estetrol†
- Estradiol
-
Estradiol esters (e.g., estradiol acetate, estradiol benzoate, estradiol cypionate, estradiol enanthate, estradiol undecylate, estradiol valerate, polyestradiol phosphate, estradiol ester mixtures (Climacteron))
- Estramustine phosphate
- Estriol
-
Estriol esters (e.g., estriol succinate, polyestriol phosphate)
- Estrogenic substances
- Estrone
-
Estrone esters
-
Ethinylestradiol#
- Hydroxyestrone diacetate
- Mestranol
- Methylestradiol
- Moxestrol
- Nilestriol
-
Prasterone (dehydroepiandrosterone; DHEA)
- Promestriene
- Quinestradol
- Quinestrol
|
| Progonadotropins |
|
|
| Antiestrogens |
ER antagonists
(incl. SERMs/SERDs) |
|
| Aromatase inhibitors |
|
| Antigonadotropins |
-
Androgens/anabolic steroids (e.g., testosterone, testosterone esters, nandrolone esters, oxandrolone, fluoxymesterone)
-
D2 receptor antagonists (prolactin releasers) (e.g., domperidone, metoclopramide, risperidone, haloperidol, chlorpromazine, sulpiride)
-
GnRH agonists (e.g., leuprorelin, goserelin)
-
GnRH antagonists (e.g., cetrorelix, elagolix)
-
Progestogens (e.g., chlormadinone acetate, cyproterone acetate, gestonorone caproate, hydroxyprogesterone caproate, medroxyprogesterone acetate, megestrol acetate)
|
| Others |
|
|
|
|
|
|---|
GnRH modulators
(incl. analogues) |
|
| Gonadotropins |
|
Others
(indirect) |
| Progonadotropins |
|
| Antigonadotropins |
-
Sex steroid agonists (via negative feedback on the HPG axis): Androgens/anabolic steroids (e.g., testosterone, nandrolone esters, oxandrolone)
-
D2 receptor antagonists (prolactin releasers) (incl., domperidone, metoclopramide, risperidone, haloperidol, chlorpromazine, sulpiride)
-
Estrogens (incl., bifluranol, estradiol, estradiol esters, ethinylestradiol, diethylstilbestrol, paroxypropione)
-
Progestogens (incl. progestins, e.g., chlormadinone acetate, cyproterone acetate, hydroxyprogesterone caproate, gestonorone caproate, medroxyprogesterone acetate, megestrol acetate)
|
|
|
|
|
|---|
| ER |
| Agonists |
-
Steroidal: 2-Hydroxyestradiol
- 2-Hydroxyestrone
- 3-Methyl-19-methyleneandrosta-3,5-dien-17β-ol
- 3α-Androstanediol
- 3α,5α-Dihydrolevonorgestrel
- 3β,5α-Dihydrolevonorgestrel
- 3α-Hydroxytibolone
- 3β-Hydroxytibolone
- 3β-Androstanediol
- 4-Androstenediol
- 4-Androstenedione
- 4-Fluoroestradiol
- 4-Hydroxyestradiol
- 4-Hydroxyestrone
- 4-Methoxyestradiol
- 4-Methoxyestrone
- 5-Androstenediol
- 7-Oxo-DHEA
- 7α-Hydroxy-DHEA
- 7α-Methylestradiol
- 7β-Hydroxyepiandrosterone
- 8,9-Dehydroestradiol
- 8,9-Dehydroestrone
- 8β-VE2
- 10β,17β-Dihydroxyestra-1,4-dien-3-one (DHED)
- 11β-Chloromethylestradiol
- 11β-Methoxyestradiol
- 15α-Hydroxyestradiol
- 16-Ketoestradiol
- 16-Ketoestrone
- 16α-Fluoroestradiol
- 16α-Hydroxy-DHEA
- 16α-Hydroxyestrone
- 16α-Iodoestradiol
- 16α-LE2
- 16β-Hydroxyestrone
- 16β,17α-Epiestriol (16β-hydroxy-17α-estradiol)
-
17α-Estradiol (alfatradiol)
- 17α-Dihydroequilenin
- 17α-Dihydroequilin
- 17α-Epiestriol (16α-hydroxy-17α-estradiol)
- 17α-Ethynyl-3α-androstanediol
- 17α-Ethynyl-3β-androstanediol
- 17β-Dihydroequilenin
- 17β-Dihydroequilin
- 17β-Methyl-17α-dihydroequilenin
- Abiraterone
- Abiraterone acetate
- Alestramustine
- Almestrone
-
Anabolic steroids (e.g., testosterone and esters, methyltestosterone, metandienone (methandrostenolone), nandrolone and esters, many others; via estrogenic metabolites)
- Atrimustine
- Bolandiol
- Bolandiol dipropionate
- Butolame
- Clomestrone
-
Cloxestradiol
- Conjugated estriol
- Conjugated estrogens
- Cyclodiol
- Cyclotriol
- DHEA
- DHEA-S
- ent-Estradiol
- Epiestriol (16β-epiestriol, 16β-hydroxy-17β-estradiol)
- Epimestrol
- Equilenin
- Equilin
- ERA-63 (ORG-37663)
- Esterified estrogens
- Estetrol
-
Estradiol
- Estramustine
- Estramustine phosphate
- Estrapronicate
- Estrazinol
-
Estriol
- Estrofurate
- Estrogenic substances
- Estromustine
-
Estrone
- Etamestrol (eptamestrol)
-
Ethinylandrostenediol
-
Ethinylestradiol
- Ethinylestriol
- Ethylestradiol
- Etynodiol
- Etynodiol diacetate
- Hexolame
- Hippulin
- Hydroxyestrone diacetate
- Lynestrenol
- Lynestrenol phenylpropionate
- Mestranol
- Methylestradiol
- Moxestrol
- Mytatrienediol
- Nilestriol
- Norethisterone
- Noretynodrel
- Orestrate
- Pentolame
- Prodiame
- Prolame
- Promestriene
- RU-16117
- Quinestradol
- Quinestrol
- Tibolone
-
Xenoestrogens: Anise-related (e.g., anethole, anol, dianethole, dianol, photoanethole)
-
Chalconoids (e.g., isoliquiritigenin, phloretin, phlorizin (phloridzin), wedelolactone)
-
Coumestans (e.g., coumestrol, psoralidin)
-
Flavonoids (incl. 7,8-DHF, 8-prenylnaringenin, apigenin, baicalein, baicalin, biochanin A, calycosin, catechin, daidzein, daidzin, ECG, EGCG, epicatechin, equol, formononetin, glabrene, glabridin, genistein, genistin, glycitein, kaempferol, liquiritigenin, mirificin, myricetin, naringenin, penduletin, pinocembrin, prunetin, puerarin, quercetin, tectoridin, tectorigenin)
- Lavender oil
-
Lignans (e.g., enterodiol, enterolactone, nyasol (cis-hinokiresinol))
-
Metalloestrogens (e.g., cadmium)
-
Pesticides (e.g., alternariol, dieldrin, endosulfan, fenarimol, HPTE, methiocarb, methoxychlor, triclocarban, triclosan)
-
Phytosteroids (e.g., digitoxin (digitalis), diosgenin, guggulsterone)
-
Phytosterols (e.g., β-sitosterol, campesterol, stigmasterol)
-
Resorcylic acid lactones (e.g., zearalanone, α-zearalenol, β-zearalenol, zearalenone, zeranol (α-zearalanol), taleranol (teranol, β-zearalanol))
-
Steroid-like (e.g., deoxymiroestrol, miroestrol)
-
Stilbenoids (e.g., resveratrol, rhaponticin)
-
Synthetic xenoestrogens (e.g., alkylphenols, bisphenols (e.g., BPA, BPF, BPS), DDT, parabens, PBBs, PHBA, phthalates, PCBs)
- Others (e.g., agnuside, rotundifuran)
|
Mixed
(SERMs) |
|
| Antagonists |
-
Coregulator-binding modulators: ERX-11
|
|
| GPER |
| Agonists |
|
| Antagonists |
|
| Unknown |
|
|
|